ExeVir Bio Forges Ahead with Unique Next Generation COVID-19 Therapy and Appoints Dr. Jeanne Bolger as Chair of the Board of Directors

ExeVir is pleased to announce today the appointment of Dr Jeanne Bolger as Chair to the Board of Directors.
Dr. Bolger has over 35 years of pharmaceutical industry experience in management roles across R&D, Commercial and Business Development and Venture Investments at Johnson & Johnson Innovation – JJDC and at GlaxoSmithKline.
ExeVir Announces First Patient Enrolled in Phase 1b/2 Clinical Study evaluating XVR011 as antiviral treatment of patients hospitalised for COVID-19

ExeVir, which is developing single domain antibody therapies providing broad protection against viral infections, today announces that the first patient has been treated in a Phase 1b/2 global clinical study of XVR011, its potent COVID-19 neutralizing antibody.
ExeVir announces first subjects dosed Phase 1 clinical study of potent COVID-19 neutralizing antibody

ExeVir, which is developing single domain antibody therapies providing broad protection against viral infections, today announces that the first subjects have been dosed in a Phase I clinical study of XVR011, its llama-derived antibody for the treatment and prevention of COVID-19.
ExeVir and Mithra announce manufacturing collaboration for innovative Covid-19 treatments

ExeVir, which is developing single domain antibody therapies providing broad protection against viral infections, and Mithra Pharmaceuticals (Euronext Brussels: MITRA) announce a new collaboration to utilize the fill and finish capabilities for ExeVir’s innovative therapies at Mithra’s integrated R&D and manufacturing platform, Mithra CDMO.
ExeVir’s COVID-19 antibody candidate neutralizes coronavirus Variants of Concern
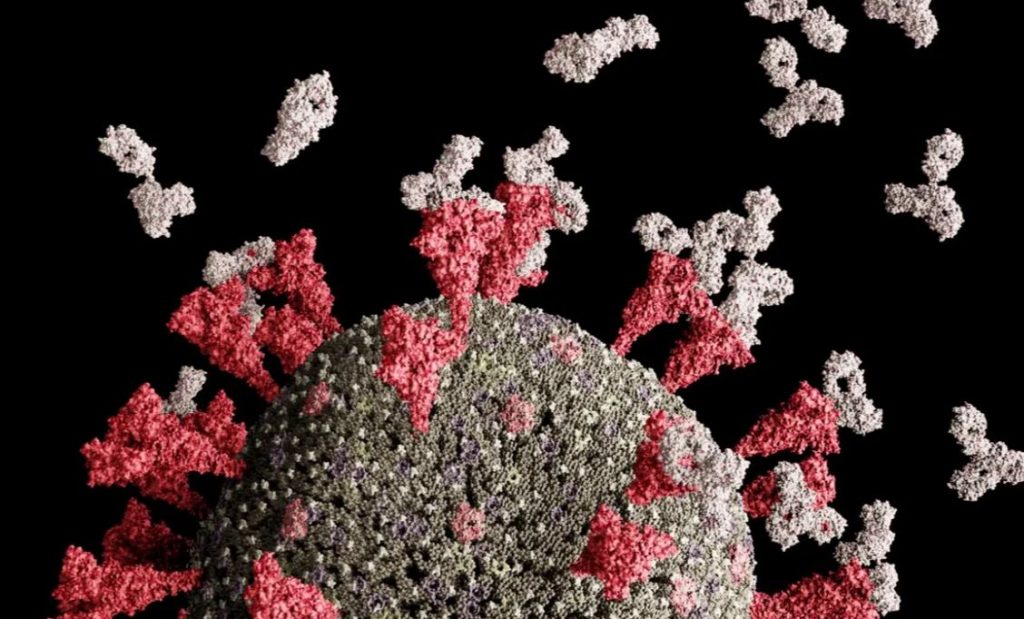
ExeVir, which is developing single domain antibody therapies providing broad protection against viral infections, today announces that it has generated data on its COVID-19 antibody XVR011 which demonstrate in vitro neutralization activity against SARS-CoV-2 Variants of Concern Gamma and Delta. The company has also obtained grant funding of €3 million from the Flanders Agency for Innovation & Entrepreneurship (VLAIO) to accelerate clinical development of lead asset, the unique llama-derived VHH72-Fc antibody XVR011.
ExeVir Announces Close of $50 million/ EUR42 million Series A Financing

ExeVir, which is developing single domain antibody therapies providing broad protection against viral infections, today announces that it has closed its Series A financing raising a total of EUR 42 million / US$50 million.
